*Part of this work was presented at the 14th Tumour Models Summit, London, December 2025.
Introduction
Preclinical models play a crucial role in the development of novel diagnostics and therapies for prostate cancer. Among these, the LNCaP cell line, derived from human prostate adenocarcinoma, is widely utilized due to its consistent tumorigenic properties and expression of prostate-specific markers [1, 2]. When implanted into immunodeficient mice, LNCaP cells form xenograft tumors, which allow detailed evaluation of tumor growth kinetics and therapeutic efficacy [2, 3].
Prostate-Specific Membrane Antigen (PSMA) is highly expressed in prostate cancer tissues and represents a well-established and valuable target for both imaging and radionuclide therapies [4, 5]. In LNCaP xenograft models, PSMA expression can be effectively visualized using radiolabeled compounds such as ¹⁷⁷Lu-labeled tracers, enabling precise assessment of tumor targeting and treatment response [6].
This study focuses on (1) establishing and comprehensively characterizing the LNCaP xenograft model, and (2) assessing in vivo the PSMA expression via two-dimensional imaging with a ¹⁷⁷Lu-labeled PSMA-targeting compound. The preclinical data generated aim to provide insights into tumor development and target engagement, facilitating the translation of radioligand therapies.
Imaging Systems
The γ-eye™, (BIOEMTECH, Greece) is a dedicated preclinical imaging scanner, specifically designed for real-time screening of SPECT and alpha particle-emitting isotopes, in clinically translational activities and scanning times. The detector consists of a 6 mm-thick pixelated GaGG:Ce scintillator (EPIC crystal, China), coupled to an array of 6 mm × 6 mm Hamamatsu silicon photomultipliers (S14160-6050HS MPPC). The scintillator array includes 58 × 116 elements with a pitch of 1.7 mm × 1.7 mm, resulting in a total active field of view (FOV) of 98.6 mm × 197.2 mm, which is sufficient for whole-body imaging of four mice, enabling high-throughput in vivo screening of labeled compounds. Featuring a small footprint (60 cm × 60 cm × 50 cm) and a light weight, it enables desktop imaging. The scanner is equipped with a series of different collimators, which are application-specific, interchangeable tungsten parallel-hole made.
For the present experiments, animals were positioned on a heated imaging bed with integrated inhalation anesthesia and imaged in real time using the VISUAL|eyes™ software (BIOEMTECH, Greece). The main photopeaks used for imaging ¹⁷⁷Lu were 113 and 208 keV.
Tumour model establishment
LNCaP cells were subcutaneously inoculated into the shoulder of two (2) NXG immunodeficient mice (NOD-Prkdcscid-IL2rgᵀᵐ¹/Rj strain). Tumor growth was monitored twice per week using a caliper, and tumor volume was calculated. Growth kinetics are shown in the tumor growth curve.
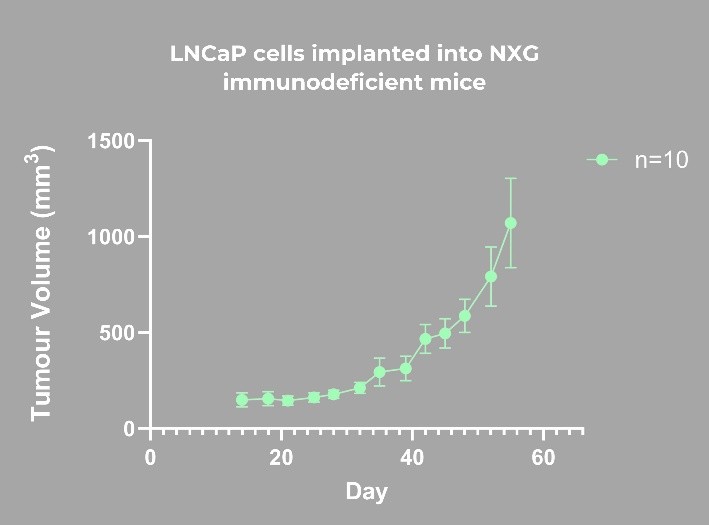
Figure 1. Tumor growth curve of LNCap cells (in PBS:Matrigel:1:1) implanted in NXG immunodeficient mice. Tumor volume was measured twice a week until reached humane endpoints.
Radiotracer administration and imaging
When tumor volume reached the target size of approximately [≈500 mm³], mice received an intravenous injection of a ¹⁷⁷Lu-labeled PSMA-targeting radiotracer (≈18 MBq). Two-dimensional (planar) imaging was performed in vivo at 1-, 4-, and 24-hours post-injection using a dedicated 2D imaging device (γ-eye™, BIOEMTECH, Greece). For each time point, two mice were imaged simultaneously within a single acquisition. The duration of each imaging session was 15 minutes. Image post-processing was performed using the VISUAL|eyes™ platform and tumor uptake was calculated for each animal.
Biodistribution study
At 24 hours post-injection, mice were euthanized and blood, kidneys, liver, lungs, spleen, muscle, tail and tumor were collected. Radioactivity in each organ was measured using a Hidex γ-counter.
Results
In vivo imaging was performed on two separate mice, labeled as 1.1 and 1.2. Tumor uptake (kBq) was quantified at 1, 4 and 24 hours post injection (p.i.) of the ¹⁷⁷Lu-PSMA-targeting radiotracer.
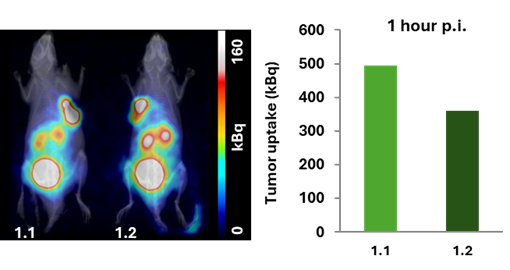
Figure 2. 1 hour p.i., imaging. Tumor uptake was 495 kBq for mouse 1.1 and 361 kBq for mouse 1.2.
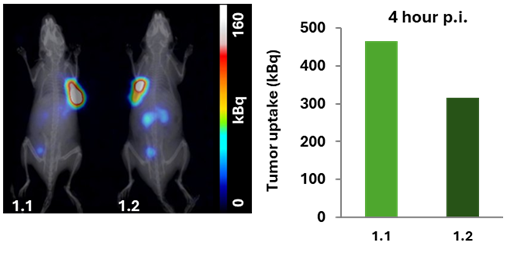
Figure 3. 4 hours p.i., imaging. Tumor uptake was 466 kBq and 316 kBq for mice 1.1 and 1.2, respectively.
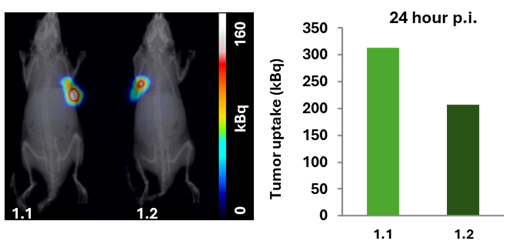
Figure 4. 24 hours p.i., imaging. Tumor uptake for mouse 1.1 was 313 kBq and 206 kBq for mouse 1.2, respectively.
Key points
- The LNCaP PSMA-positive xenograft model was successfully established, generating stable tumors suitable for radiotracer evaluation.
- The study confirms the uptake of the PSMA-targeted compound in LNCaP tumors, which highly express PSMA.
- At 1-hour post-injection, tumor uptake reached 495 kBq for animal 1.1 and 361 kBq for animal 1.2 and decreased to approximately 466 kBq and 316 kBq at 4 hours, respectively. By 24 hours p.i., tumor uptake had decreased (313 kBq for animal 1.1 and 206 kBq for animal 1.2), following overall radiotracer clearance. These findings were consistent with the biodistribution results (302 kBq for animal 1.1 and 223 kBq for animal 1.2). Despite some inter-animal variability, both mice showed clear PSMA-specific uptake, supporting the robustness and reproducibility of the LNCaP model.
- The combined imaging and biodistribution data highlight the model’s suitability for assessing PSMA-targeted cold compounds, radioligand therapies and dose – response relationships.
References
- Devlies W et al., Cancers (Basel), 2021;13:915.
- Abate-Shen C, Nunes de Almeida F. Cancer Res., 2022;82:1689-1691.
- Liu W et al., BMC Cancer, 2022;22:173.
- Kiess AP et al., Nucl Med Mol Imaging, 2015;59:241-268.
- Bouchelouche K et al., Discov Med, 2010;9:55-61.
- Ruigrok EAM et al., Eur J Nucl Med Mol Imaging, 2021;48:1339-1350.
Please note that animal care and use was conducted at an authorized user establishment in compliance to European legislation on the protection of animals used for scientific purposes (Directive 2010/63/EU).